Articles | March 26th, 2026
Ideal pH Ranges and How They Affect Nutrient Uptake
By Nicholas Peacock
Introduction
If you have ever fed your plants the right amount of fertilizer and still watched them struggle, there is a good chance the problem was not the nutrients themselves. It was the pH of your growing medium. Soil and substrate pH plays an enormous role in determining whether the nutrients you apply actually become available to plant roots. Get the pH wrong, and even the most carefully balanced feeding program will fall short.
This article breaks down what pH really means for your plants, which ranges work best for different growing situations, and how shifts in pH can lock out critical nutrients or release them in toxic amounts. Whether you grow in soil, coco coir, or a hydroponic system, understanding pH is one of the simplest and most impactful things you can do to improve plant health and yields.

What Is pH and Why Does It Matter?
pH is a measure of how acidic or alkaline a solution is, expressed on a scale from 0 to 14. A reading of 7.0 is considered neutral. Anything below 7.0 is acidic, and anything above 7.0 is alkaline (also called basic). Most plants prefer a slightly acidic environment, somewhere between 5.5 and 7.0 depending on the species and the growing medium.
What many growers do not realize is that pH does not just describe the character of your soil or nutrient solution. It directly controls the chemical availability of every essential nutrient. At certain pH levels, nutrients dissolve easily and flow right into the root system. At other pH levels, those same nutrients react with minerals in the soil and form compounds that roots simply cannot absorb. This phenomenon is known as nutrient lockout, and it is one of the most common reasons plants show deficiency symptoms even when plenty of fertilizer has been applied.
Ideal pH Ranges by Growing Medium
The ideal pH range depends heavily on whether you are growing in soil, soilless media like coco coir or peat, or a purely hydroponic setup. Each medium interacts with nutrients differently, and so each one has a slightly different sweet spot.
| Growing Medium | Ideal pH Range | Notes |
| Mineral Soil | 6.0 – 7.0 | Most versatile range; suits the majority of vegetables and flowers |
| Coco Coir / Peat | 5.5 – 6.5 | Slightly lower target due to reduced buffering capacity |
| Hydroponics (inert media) | 5.5 – 6.0 | Tighter range needed because there is no soil buffer |
| Acid-loving plants (blueberries, azaleas) | 4.5 – 5.5 | These species have evolved to thrive in acidic conditions |
In soil, the natural buffering capacity of clay, organic matter, and mineral particles helps keep pH relatively stable. That is why the acceptable range in soil is a bit wider. In hydroponic systems, there is almost no buffer at all, so even small changes in pH can have immediate effects on nutrient availability. That is why hydroponic growers tend to monitor and adjust pH much more frequently.
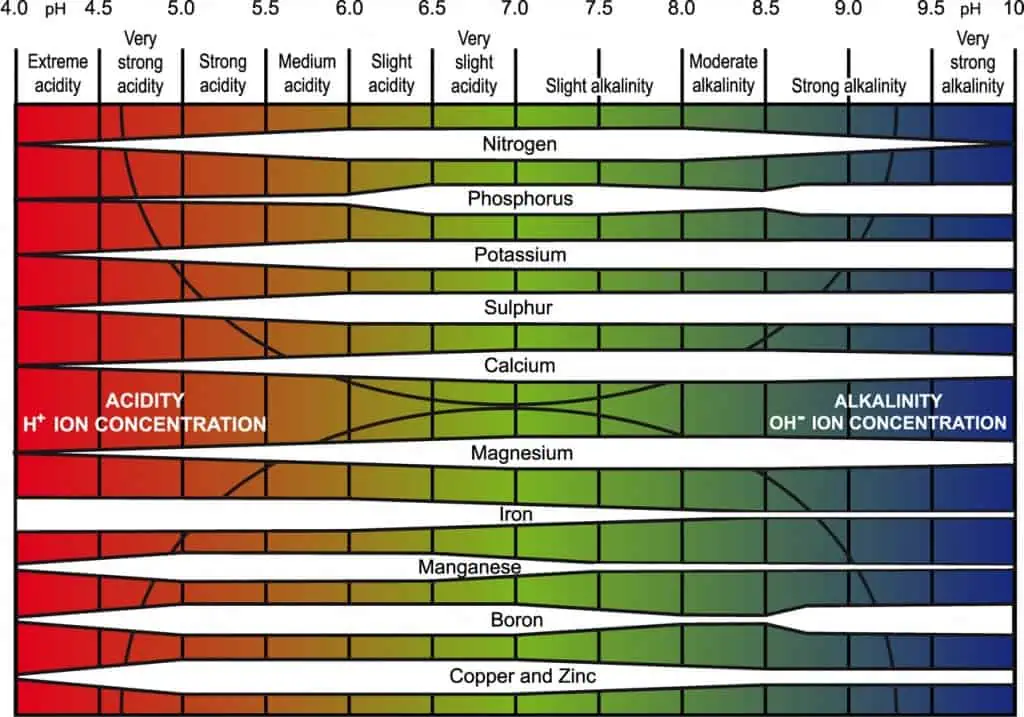
How pH Affects Individual Nutrients
Each nutrient has its own solubility curve, meaning it is most available at certain pH levels and least available at others. Here is how pH shifts tend to affect the major groups of plant nutrients.
Primary Macronutrients (N, P, K)
Nitrogen, phosphorus, and potassium are the nutrients plants consume in the largest quantities. Nitrogen is most available between pH 6.0 and 8.0, so it remains accessible across a fairly wide range. Phosphorus, on the other hand, is much pickier. It reaches peak availability between about 6.0 and 7.0. When pH drops below 5.5, phosphorus binds with iron and aluminum. When pH climbs above 7.5, it locks up with calcium. Potassium stays reasonably available across a broad range, but uptake can be hindered in very acidic soils where other competing ions dominate the root exchange sites.
Secondary Macronutrients (Ca, Mg, S)
Calcium and magnesium are most available in slightly acidic to neutral conditions, roughly pH 6.0 to 7.5. If your soil drifts too far below 6.0, calcium uptake drops off significantly, which can lead to problems like blossom end rot in tomatoes and peppers. Sulfur availability is fairly stable, though it tends to decrease slightly in very alkaline soils.
Micronutrients (Fe, Mn, Zn, Cu, B, Mo)
This is where pH management gets especially interesting. Iron, manganese, zinc, copper, and boron all become more available as pH drops. In acidic conditions below 5.5, these micronutrients can actually become too available, reaching levels that are toxic to plant tissue. This is a common problem in overly acidic soils where manganese or aluminum toxicity damages root systems and stunts growth.
Molybdenum is the odd one out among the micronutrients. Unlike the others, it actually becomes more available as pH rises. Plants growing in very acidic soils sometimes show molybdenum deficiency even when the element is present in adequate amounts, simply because the low pH prevents them from absorbing it.
The Lockout Zone: What Happens When pH Drifts
When pH moves outside the ideal range, you do not just lose access to one nutrient. Multiple nutrients can become unavailable at the same time, creating a cascade of deficiency symptoms that can be confusing to diagnose. A plant growing at pH 4.5, for instance, might show phosphorus deficiency, calcium deficiency, and manganese toxicity all at once. Meanwhile, a plant stuck at pH 8.0 could display iron chlorosis (yellowing between the veins of new leaves), zinc deficiency, and boron deficiency simultaneously.
This is why experienced growers always check pH before they start adding extra nutrients. If the pH is off, adding more fertilizer will not solve the problem. It will just waste product and potentially make the situation worse by increasing salt buildup around the roots.
Practical Tips for Managing pH
Keeping pH in the right range does not have to be complicated, but it does require some attention. Here are some straightforward strategies that work across different growing setups.
Test regularly. A simple pH pen or test kit is one of the best investments a grower can make. For soil, test the runoff water that drains from the bottom of your pots. For hydroponics, test your reservoir daily. Small corrections are always easier than big ones.
Adjust gradually. Use pH up (potassium hydroxide or potassium carbonate) or pH down (phosphoric acid or citric acid) solutions to nudge the pH in the right direction. Make small adjustments and retest. Overshooting in either direction just creates new problems.
Use appropriate amendments in soil. If your soil pH is too low, agricultural lime (calcium carbonate) is the standard fix. If pH is too high, elemental sulfur or an acidifying fertilizer like ammonium sulfate can bring it down over time. Keep in mind that soil amendments work slowly, often taking weeks or months to show full effect.
Consider your water source. Tap water in many regions is naturally alkaline, sometimes above pH 8.0. If you are using tap water without adjusting it first, you may be slowly pushing your growing medium toward a pH that locks out key nutrients. Always test and adjust your water before mixing in fertilizers.
Allow slight pH drift in hydroponics. Some experienced hydroponic growers let their solution pH swing gently between 5.5 and 6.5 rather than chasing a single number. This approach allows different nutrients to reach peak availability at different times as the pH naturally fluctuates, and it can actually improve overall nutrient uptake compared to holding pH at one fixed point.
Conclusion
pH might seem like a small detail, but it is really the gatekeeper for everything your plants eat. No amount of premium fertilizer will help if the pH of your root zone is locking out the nutrients your plants need most. By understanding the ideal ranges for your growing medium, knowing which nutrients are sensitive to pH shifts, and testing regularly, you can avoid a huge percentage of the nutrient problems that plague both beginner and experienced growers.
The good news is that managing pH is not expensive or difficult. A reliable pH meter, a basic understanding of how your nutrients interact with your growing medium, and a willingness to test and adjust consistently are all you need. Get the pH right, and your plants will thank you with stronger growth, healthier foliage, and better harvests.
